CONTENTS
INTRODUCTION 1)
USES OF AVOGADRO CONSTANT 3)
METHODS USED TO OBTAIN AVOGADRO CONSTANT 4)
PROCEDURE 8)
OBSERVATIONS 10)
PRECAUTIONS 11)
BIBLIOGRAPHY 12)

Amedeo Avogadro
In chemistry and physics, the Avogadro constant (symbols: L, NA), also called Avogadro’s number, is the number of “elementary entities” (usually atoms or molecules) in one mole, that is, the number of atoms in exactly 12 grams of carbon-12.The 2006 CODATA recommended value is:
NA = 6.02214179(30) mol-1
The Avogadro constant is named after the early nineteenth century Italian scientist Amedeo Avogadro, who, in 1811, first proposed that the volume of a
1) gas at a given pressure and temperature is proportional to the number of atoms or molecules regardless of the nature of the gas. The French physicist Jean Perrin in 1909 proposed naming the constant in honour of Avogadro.
The value of the Avogadro constant was first indicated by Johann Josef Loschmidt who, in 1865, estimated the average diameter of the molecules in air by a method that is equivalent to calculating the number of particles in a given volume of gas. This latter value, the number density of particles in an ideal gas, is now called the Loschmidt constant in his honour, and is approximately proportional to the Avogadro constant.
Jean Perrin originally proposed the name “Avogadro’s number” (N) to refer to the number of molecules in one gram-molecule of oxygen. The change in name to “Avogadro constant” (NA) came with the introduction of the mole as a separate base unit in the International System of Units (SI) in 1971, which recognized amount of substance as an independent dimension of measurement. With this recognition, the Avogadro constant was no longer a pure number but a physical quantity associated with a unit of measurement, the reciprocal mole (mol−1) in SI units.
2) Because of its role as a scaling factor, the Avogadro constant provides the link between a number of useful physical constants when moving between the atomic scale and the macroscopic scale. For example, it provides the relationship between:
The gas constant R and the Boltzmann constant kB:
R = kB NA = 8.314472(15) J mol-1 K-1
The Faraday constant F and the elementary charge e:
F = NAe = 96485.3389(83) C mol-1
The Avogadro constant also enters into the definition of the unified atomic mass unit, u:
1u = Mu = 1.660538782(83) x 10-24 g
NA
where Mu is the molar mass constant.
Coulometry

The earliest accurate method to measure the value of the Avogadro constant was based on coulometry. The principle is to measure the Faraday constant, F,
4) which is the electric charge carried by one mole of electrons, and to divide by the elementary charge, e, to obtain the Avogadro constant.
NA = Fe
The classic experiment is that of Bowers and Davis at NIST, and relies on dissolving silver metal away from the anode of an electrolysis cell, while passing a constant electric current I for a known time t. If m is the mass of silver lost from the anode and Ar the atomic weight of silver, then the Faraday constant is given by:
![]()
Their value for the conventional Faraday constant is F90 = 96 485.39(13) C/mol, which corresponds to a value for the Avogadro constant of 6.022 1449(78) × 1023 mol−1: both values have a relative standard uncertainty of 1.3 × 10–6.
Electron around nucleus
5) The CODATA value for the Avogadro constant is determined from the ratio of the molar mass of the electron Ar(e)Mu to the rest mass of the electron me:
![]()
![]()
The “relative atomic mass” of the electron, Ar(e), is a directly-measured quantity, and the molar mass constant, Mu, is a defined constant in the SI system. The electron rest mass, however, is calculated from other measured constants.
The main limiting factor in the precision to which the value of the Avogadro constant is known is the uncertainty in the value of the Planck constant, as all the other constants which contribute to the calculation are known much more precisely.
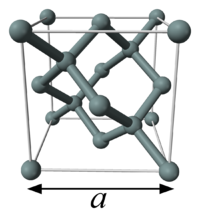
X Ray Electron mass method (CODATA)
Ball-and-stick model of the unit cell of silicon.
One modern method to calculate the Avogadro constant is to use ratio of the molar volume, Vm, to the unit cell volume, Vcell, for a single crystal of silicon:
6)
![]()
The factor of eight arises because there are eight silicon atoms in each unit cell.
The unit cell volume can be obtained by X-ray crystallography; as the unit cell is cubic, the volume is the cube of the length of one side. The isotope proportional composition of the sample used must be measured and taken into account.
Silicon occurs with three stable isotopes – 28Si, 29Si, 30Si – and the natural variation in their proportions is greater than other uncertainties in the measurements.
The atomic weight Ar for the sample crystal can be calculated, as the relative atomic masses of the three nuclides are known with great accuracy. This, together with the measured density ρ of the sample, allows the molar volume Vm to be found by:
![]()
where Mu is the molar mass constant. The 2006 CODATA value for the molar volume of silicon.
As of the 2006 CODATA recommended values, the relative uncertainty in determinations of the Avogadro constant by the X-ray crystal density method is 1.2 × 10–7, about two and a half times higher than that of the electron mass method.
PROCEDURE
Materials
- Direct current source (battery or power supply)
- Insulated wires and possibly alligator clips to connect the cells
- 2 Electrodes (e.g., strips of copper, nickel, zinc, or iron)
- 250-ml beaker of 0.5 M H2SO4 (sulphuric acid)
- Water
- Alcohol (e.g., methanol or isopropyl alcohol)
- Small beaker of 6 M HNO3 (nitric acid)
- Ammeter or multimeter
- Stopwatch
- Analytical balance capable of measuring to nearest 0.0001 gram
Obtain two copper electrodes. Clean the electrode to be used as the anode by immersing it in 6 M HNO3 in a fume hood for 2-3 seconds. Remove the electrode promptly or the acid will destroy it. Do not touch the electrode with your fingers. Rinse the electrode with clean tap water. Next, dip the electrode into a beaker of alcohol. Place the electrode onto a paper towel. When the electrode is dry, weigh it on an analytical balance to the nearest 0.0001 gram.
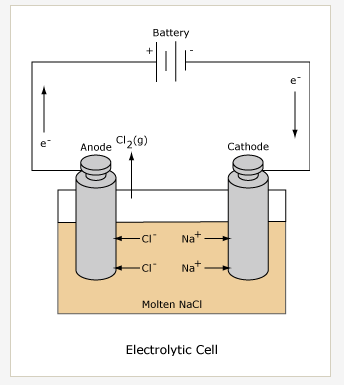
The apparatus looks superficially like this diagram of an electrolytic cell
Notice that we are using two beakers connected by an ammeter rather than
8) having the electrodes together in a solution. Take beaker with 0.5 M H2SO4 and place an electrode in each beaker. Before making any connections be sure the power supply is off and unplugged. The power supply is connected to the ammeter in series with the electrodes. The positive pole of the power supply is connected to the anode. The negative pin of the ammeter is connected to the anode. The cathode is connected to the positive pin of the ammeter. Finally, the cathode of the electrolytic cell is connected to the negative post of the battery or power supply. Remember, the mass of the anode will begin to change as soon as you turn the power on, so have your stopwatch ready!
You need accurate current and time measurements. The amperage should be recorded at one minute (60 sec) intervals. Be aware that the amperage may vary over the course of the experiment due to changes in the electrolyte solution, temperature, and position of the electrodes. The amperage used in the calculation should be an average of all readings. Allow the current to flow for a minimum of 1020 seconds (17.00 minutes). Measure the time to the nearest second or fraction of a second. After 1020 seconds turn off the power supply record the last amperage value and the time.
Now you retrieve the anode from the cell, dry it as before by immersing it in alcohol and allowing it to dry on a paper towel, and weigh it. If you wipe the anode you will remove copper from the surface and invalidate your work!
OBSERVATIONS
The following observations were made:
Anode mass lost: 0.3554 grams (g)
Current(average): 0.601 amperes (amp)
Time of electrolysis: 1802 seconds (s)
Remember:
one ampere = 1 coulomb/second or one amp.s = 1 coul
charge of one electron is 1.602 x 10-19 coulomb
- Find the total charge passed through the circuit.
(0.601 amp)(1 coul/1amp-s)(1802 s) = 1083 coul - Calculate the number of electrons in the electrolysis.
(1083 coul)(1 electron/1.6022 x 1019coul) = 6.759 x 1021 electrons - Determine the number of copper atoms lost from the anode.
The electrolysis process consumes two electrons per copper ion formed. Thus, the number of copper (II) ions formed is half the number of electrons.
Number of Cu2+ ions = ½ number of electrons measured
Number of Cu2+ ions = (6.752 x 1021 electrons)(1 Cu2+ / 2 electrons)
Number of Cu2+ ions = 3.380 x 1021 Cu2+ ions - Calculate the number of copper ions per gram of copper from the number of copper ions above and the mass of copper ions produced.
The mass of the copper ions produced is equal to the mass loss of the anode. (The mass of the electrons is so small as to be negligible, so the mass of the copper (II) ions is the same as the mass of copper atoms.)
mass loss of electrode = mass of Cu2+ ions = 0.3554 g
3.380 x 1021 Cu2+ ions / 0.3544g = 9.510 x 1021 Cu2+ ions/g = 9.510 x 1021 Cu atoms/g
- Calculate the number of copper atoms in a mole of copper, 63.546 grams.
Cu atoms/mole of Cu = (9.510 x 1021 copper atoms/g copper)(63.546 g/mole copper)
Cu atoms/mole of Cu = 6.040 x 1023 copper atoms/mole of copper
This is my measured value of Avogadro’s number! - Calculate percent error.
Absolute error: |6.02 x 1023 – 6.04 x 1023 | = 2 x 1021
Percent error: (2 x 10 21 / 6.02 x 10 23)(100) = 0.3 %
Precautions
- The chemicals should be handled with care to avoid any mishaps.
- Do not switch on the battery before you have setup the entire circuit.
- Be accurate while starting and stopping the stopwatch.
- Do not wipe the anode.
BIBLIOGRAPHY
- http://www.iupac.org/goldbook/A00543.pdf
- http://gemini.tntech.edu/%7Etfurtsch/scihist/avogadro.htm
- http://www.americanscientist.org/issues/pub/2007/2/an-exact-value-for-avogadros-number
- http://www.inrim.it/Nah/Web_Nah/home.htm
You can find other Chemistry Projects here.
