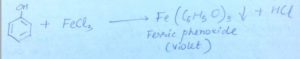Nature of Money and evolution
Exchange is a way of life and money is an instrument that facilitates exchange. Hence, we can say that nature of money is one that facilitates exchange.
Before money came into being, goods were exchanged for goods. This system is known as barter system of exchange. However, barter system proved to be inefficient considering the increased wants and greater need of exchange. Therefore, money was invented as a solution to the problem of not having a common medium of exchange.
Metal coins of gold and silver were the first to come followed by alloy coins and finally the paper currency. The 21st century saw the introduction of plastic money in form of debit cards and credit cards.
Barter system and its drawbacks
It is an exchange system in which goods are exchanged for goods. For example, I am a dairy producer who sells milk and I need a pair of shoes. I would search for a person to whom I can sell milk in return of a pair of shoes. This is also known as double coincidence of wants. This means that my want of shoes must coincide with somebody’s want of milk. As you can see in the diagram, needs of two persons should coincide or match for barter system to work.
An economy where such system of exchange works is known as a C-C economy. A C-C or Commodity to Commodity economy is one which is influenced by Barter system of exchange. Hence you can see that Barter System, double coincidence of wants and C-C economy are all related concepts.
The Barter system had its own drawbacks as well. If you remember I asked you to think over this question earlier. As you can see in the diagram, these are the four major drawbacks. We will talk about each of these drawbacks in detail now.
Difficulty of double coincidence of wants
For double coincidence of wants to exist, wants of two individuals should coincide. This possesses a major problem as you cannot always find a suitable person who wants your shirt and at the same time has rice that you want to buy. Hence, money was invented to solve this problem.
Lack of common unit of value
Under barter system, goods are valued against goods. For example, a bike would be valued against shoes. Or rice would be valued against a cow. This makes exchange difficult. With no common unit of value, it was difficult to estimate prices of products. Money provided that common unit value and a better financial accounting system.
Lack of system for future payments
In a barter system, it is difficult to do such payments which are to be given in the future. It is also problematic when payments for any service has to be made. For example, I hire a person to repair my tap. Now, it is difficult to conclude payments in the form of goods for the service he offered.
Money solved this problem as there is an option to pay the service man at a later date and time.
Lack of system for storage and transfer of value
Since there was no money in the C-C Economy, wealth was stored and saved in the form of goods which was a problematic issue. Moreover, transfer of wealth from one individual to another also was a herculean task. Evolution of money made storage and transfer easy.
Defining Money
Money can be described something that:
- acts as an instrument of exchange
- is a store of value
- is a measure of value
- a standard for deferred payments
These are the four major components of the definition of money. Basically, money performs or has the ability to perform these functions.
We can also define money from a narrow and broad perspective. As per the narrow concept, things which act as money will be included in money. For example, all the coins, paper currency and even cheques and deposits will constitute money.
As per the broad concept, something that can be converted into money at a later date is also included in the definition of money. These include term or fixed deposits. So, both money items and future money items (which can be converted into money at later date) together constitute broad definition of money.
Moving on, now let’s talk about fiat money and fiduciary money. Money issued by the government authority is known as fiat money. Any Currency will be an example for this
On the other hand, money which is backed up by trust between payer and payee is known as fiduciary money. There is no issuing or ordering authority in this case. Cheque is an example for this. They work on the principle of trust between payer and payee.
Now let’s get a bit technical and understand the difference between money value of money and commodity value of money. Money value of money is something which is inscribed or printed on the money. For example, money value money of a 10 rupee note is 10 as that is printed over it.
Whereas commodity value of money is the intrinsic value of money or the value of material used in making the money. For example, value of steel in making a coin will be known as commodity value of money.
And finally, we will understand the difference between full bodied money and credit money. Full Bodied money is when the money value (value printed on the currency) is equal to the commodity value (value of the material of which the currency is made). On the other hand, credit money is when the money value is more than the commodity value.
Functions of Money
Functions of money can be broadly divided into four parts:
- Primary functions
- Secondary functions
- Static Functions and
- Dynamic functions
Primary Functions
1. Medium of exchange
Money acts as a medium of exchange between two individuals. For example, I can buy a good or a service by paying a certain amount for it. This allows for greater exchange of goods and services which was not the case in Barter System. Introduction of Money has also separated the acts of sale and purchase due to which the economic activities have increased.
2. Measure of Value
Earlier, value of a good was valued in terms of other good (in the barter system). Now, each good is valued in terms of money which means money serves as a measure of value. This is also known as unit of account which means that value of each good is measured in monetary terms. Exchange becomes easier considering money acts as a common denominator of value. For example, I can buy shoes or shirts by paying a certain amount.
Secondary Functions
1. Deferred Payments
Payments which are made in the future are referred to as deferred payments. Money has allowed for deferred payments to be made. Now, we can use a service or buy a product and pay for it later. One can also borrow money and pay it at a later date. This promotes economic activities at a faster pace.
2. Store of Value
Money allows storage of wealth or value conveniently which was not possible in the Barter system. This is possible because Money has the feature of general acceptability, is secure and does not require much space. This is also known as asset function of money. This also means that money can be used for investment and business purposes.
3. Transfer of Value
Transferring wealth has become easier because of money. For example, I can transfer money to another person allowing him to complete a purchase at a far-off place. This feature of money has allowed for increased economic activities considering purchasing power now can be transferred easily.
Static Functions
These include those functions which ONLY help in regulating the economy of a country. They do not aim for fostering growth and development. These include both primary and secondary functions (which we have already discussed).
Dynamic Functions
These are the functions which help in improving growth and development in the economy. For example, any step by the government in either raising or decreasing the purchasing power will constitute as a dynamic function.
Supply of Money Concept
Supply of Money basically means the amount of money held by all the people of a country at a point of time. This is a stock concept and not a flow concept as it is concerned only with the money held for a particular period of time.
This concept does not include the government or the suppliers of money and only individuals who deal with money.
After we know what supply of mean, I think it’s important to know about the suppliers of money in the economy.
These are the three main suppliers of money who regulate the supply of money in the economy:
- Government
- Central Bank ( Reserve Bank of India)
- Other commercial banks
Accordingly, these are not included while measuring supply of money.
Moving on, now we will discuss about measurement of Supply of Money. There are 4 ways of measuring it:
The first way of measuring supply of money is by combining the public currency, deposits with the bank and other deposits. As you can see in the slide, formula for M1 measurement is:
M1= C+DD+OD
- ‘C’ here refers to currency which the public has like coins and notes.
- ‘DD’ refers to demand deposits with the banks.
- ‘OD’ means other deposits which includes deposits of foreign banks, foreign governments and international financial institutions with the Reserve Bank of India
These are the formulas for m2 measurment, M3 measurment and M4 measurment
M2= M1+ Deposits of public with Post Office Saving Account
M3= M1+ Net Time Deposits (Deposits for fixed term like Fixed Deposits) with the commercial Banks
M4= M3+ Total Deposits with Post Office
M1 and M2 measure are narrower in approach whereas M3 and M4 are broader in approach while calculating the money supply.
You need to remember the M1 measurement as that is the basis for all other measurements.
You can also find Class XII Notes for for Business Studies, Accounts, Maths and Economics.